


Introduction While Artificial Intelligence (AI) promises transformative efficiency in various fields, its role in pharmacovigilance
by: Upendra Shukla
As businesses increasingly move to the cloud, Software as Service (SaaS) platforms are quickly becoming
by: Prashant Guleria
Medical literature is an important source of information for suspected adverse reaction case reports of
by: Deepak Bhatia
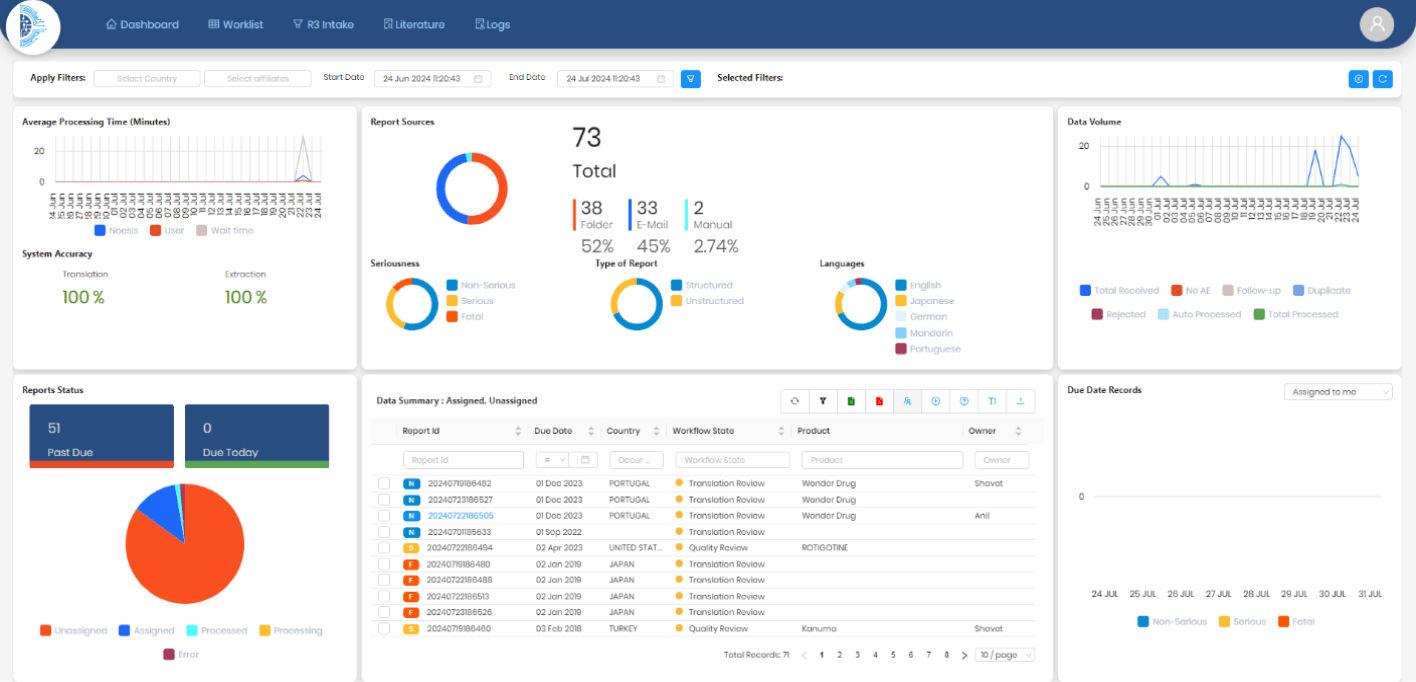
Drogevate’s NOESIS automation platform helps organizations automate the safety case management process. The Drogevate NOESIS
by: Upendra Shukla
NOESIS by Drogevate is an advanced automation platform designed to transform drug safety case management
Discover NOESIS by Drogevate, the future of drug safety case automation. Designed for pharmaceutical companies